Experiments or measurements that can be carried out using the initial suite of beamlines fall into four main categories:
- Diffraction/scattering for crystallography.
- Spectroscopy for analysis of chemical composition and speciation in the bulk material and at surfaces, down to nanometre level.
- Polarimetry for measuring the shape of complex molecules, such as proteins, and the properties of magnetic materials.
- Imaging from highly detailed imaging of small animals, and ultimately humans, down to the substructure of biological and physical materials, using light from infrared through to hard x-rays.
Some of the major common techniques employed at synchrotron facilities are:
- Protein crystallography
- X-ray diffraction
- X-ray scattering
- X-ray absorption spectroscopy
- Photo-electron spectroscopy
- Infrared spectroscopy
- Circular dichroism
- X-ray microprobe
Protein Crystallography
This is a single-crystal x-ray diffraction technique specifically for measuring the diffraction patterns of protein crystals in order to determine their structure. The Nobel Prize in Chemistry in 2006 was awarded to Prof. Roger Kornberg of Stanford University for his work in determining the interactions of RNA, which relied heavily on the use of synchrotron protein crystallography. Protein crystallography is used in drug design, as understanding the structure of proteins and other large biological molecules allows researchers to tailor pharmaceuticals to interact with them.
X-ray Diffraction (XRD)
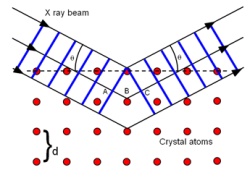
X-ray diffraction is the general term for a number of different techniques that have applications in materials science, forensics, the electronics industry, biological studies, etc. All rely on the same basic principle: x-rays are deflected by the atoms in matter and produce an interference (or diffraction) pattern. When the atoms have long-range order, the pattern produced is well-defined.
X-ray diffraction of powders is a technique that is commonly used in many research laboratories. A synchrotron has much higher flux than a laboratory source, which means that smaller amounts of material can be measured and on a faster timescale. This allows techniques such as surface diffraction and thin film diffraction to be performed, extending down to a sub-monolayer of atoms.
Synchrotron radiation is also tuneable, that is, the energy (or wavelength) of the x-rays can be changed. In the case of x-ray diffraction, this allows researchers to capture diffraction patterns at different energies which may be close to an absorption edge of an element of interest within the material being studied. This serves to highlight the specific element and is called anomalous diffraction.
X-ray Scattering (SAXS, WAXS, XRR)
X-ray scattering is the general term for the deflection of x-rays by matter, of which x-ray diffraction is but a subset. Materials with no long-range atomic order will still scatter x-rays. The smaller the scattering angle, the larger the structure that is causing the scattering.
This gives rise to two complementary techniques, SAXS and WAXS (small-angle and wide-angle x-ray scattering, respectively). As in the case of x-ray diffraction, these can be performed at selected energies to highlight an element of interest. SAXS is widely used to study materials with semi-periodic structures on the order of 1-100 nm, for example, biological assemblies, nanoparticles, quantum dots, fibres, nanoporous materials, etc. WAXS can be used to study materials with structures of the order of 0.1-1 nm, for example amorphous materials, biological molecules, clays, etc.
X-ray reflectivity is another x-ray scattering technique that relies on known theory of how x-rays penetrate into matter. It is useful for studying thin films and layered structures.
X-ray Absorption Spectroscopy (XAS, XANES, EXAFS)
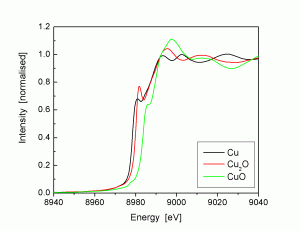
X-ray absorption spectroscopy (XAS) is an umbrella term for a range of techniques. In contrast to crystallography techniques, it is a local probe tuned to a specific element by scanning the energy (or wavelength) across an x-ray absorption edge of the element to be studied. Since it is a local probe, the technique does not depend on the material having long-range order, so it can be used to study amorphous systems and liquids (as well as systems with crystallographic order). Some of the major x-ray absorption spectroscopy techniques are:
X-ray absorption near edge structure (XANES). In the region of the absorption edge, small shifts in the edge energy are observed due to different valences of the target element. In addition, the local co-ordination of the element perturbs the edge shape. By comparing with standards with known valence and co-ordination, the local structure of the sample can be determined.
Extended x-ray absorption fine structure (EXAFS). Above the x-ray absorption edge, oscillations are observed in the energy scan due to the interaction of the ejected electron with the neighbouring atoms. These oscillations can be analysed and compared with model systems and standards to yield further information about the local structure, such as the number and type of neighbours and their distance from the target element atoms. There are several extensions to EXAFS, such as SEXAFS (surface EXAFS, which is surface-sensitive), NEXAFS (near-edge EXAFS, which includes the near-edge structure in the analysis), REFLEXAFS (reflection EXAFS), etc.
Photo-electron spectroscopy (XPS, PES, ARPES)
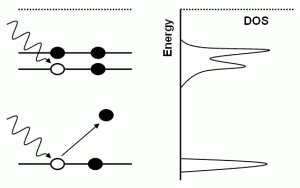
X-ray photoelectron spectroscopy (XPS, or PES) is a physical technique used at low x-ray energies to probe the binding energy of electrons within a system. This yields information about the chemical species present in the material. X-rays with an energy just above the binding energy will eject electrons, which are detected. The local atomic structure around the target element perturbs the signal, and by comparing with known substances, the chemical bonding in the material can be determined. This technique is used in surface science applications, manufacturing, etc.
Angle-resolved PES (ARPES) is a technique that separates the energy and momentum of the ejected electrons and is used for probing the density of states of materials.
Infrared spectroscopy
While infrared spectroscopy is a common laboratory technique for chemical identification, primarily of organic molecules, using a synchrotron source has several advantages.
The higher flux of a synchrotron source allows dilute samples to be measured on a shorter timescale, a situation that with a laboratory instrument would encounter problems with the signal-to-noise resolution of the detector. The wavenumber range extends over a greater range than with laboratory sources (e.g. globar) due to the tuneable nature of synchrotron radiation.
Infrared imaging can also be performed, which gives a 2D or even 3D picture of the chemical composition of the material down to micron resolution.
Circular dichroism
Circular dichroism uses polarised light to probe the structure, or ‘handedness’, of asymmetric molecules. The tuneable nature of the wavelength and polarisation of the light afforded by a synchrotron allows various aspects to be investigated. This technique is used particularly in studying biological molecules, protein folding, etc.
Circularly polarised x-rays can be used to study magnetic materials, as the x-rays interact differently with the spin-up and spin-down electrons near the Fermi level, the difference in which gives rise to magnetism.
X-ray microprobe
This is an umbrella term for microscopy using a one of the techniques listed above. A synchrotron x-ray or light beam has low divergence and can be focused to a spot that is only a few microns in size, which is then scanned in a raster fashion across the sample to build up a picture of different regions of the sample and what the diffraction pattern, x-ray absorption signal, infrared spectrum, etc. from each position is.